New article by Danielle Slomberg, Ollivier, P., Miche, H., Angeletti, B., Bruchet, A., Philibert, M., Brant, J., Labille, J., on "Nanoparticle stability in lake water shaped by natural organic matter properties and presence of particulate matter" in Science of the Total Environment
Abstract
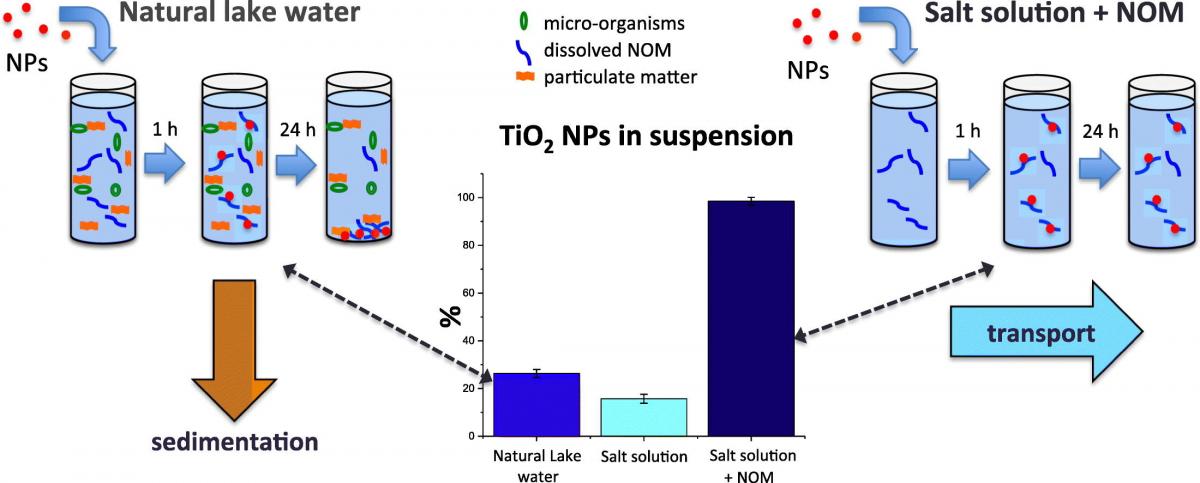
Predicting nanoparticle (NP) fate in the environment continues to remain a challenge, especially for natural sur- face water systems, where NPs can hetero-aggregate with natural organic and mineral suspended matter. Here we present the interactions and aggregation behavior of TiO2 NPs with natural organic matter (NOM) in a natural lake water. NP fate in a synthetic water of the same pH and ionic composition was also tested in the presence and absence of NOM analogs to gain insight into the different stabilizing effects of each NOM type. Several comple- mentary analytical techniques were utilized to assess lake NOM composition, including pyrolysis−gas chromatography–mass spectrometry, gel permeation chromatography, the polarity rapid-assessment method, and Nanoparticle Tracking Analysis. In the natural lake water, the TiO2 NPs preferentially interacted with mostly anionic NOM of high and medium molecular weight (~1200–1450 and 400–520 Da). Specifically, strong interac- tions with proteins and polyhydroxy aromatics were observed. NP fate and stability were determined in both raw lake water containing mineral particulate matter and total NOM (NOMtot) and filtered lake water containing only NOM b0.8 μm (NOMb0.8), with different aggregation profiles observed over time. Additionally, three times the number of TiO2 NPs remained in suspension when only NOMb0.8 was present compared to the unfiltered water containing mineral particulate matter and NOMtot. These results demonstrate the contrasting NP fates in the aquatic environment according to the presence of NOMtot vs. NOMb0.8 and further suggest that the use of pure NOM analogs may not accurately represent NP interactions and fate in the natural system.
https://doi.org/10.1016/j.scitotenv.2018.11.279